
Understanding the melting point of copper is crucial for its application in various fields. It defines the temperature range at which copper retains its structural integrity and does not transition into a liquid state. In this guide, we will explore the basic aspects of copper’s melting point, factors influencing it, and its thermal characteristics. Let’s begin:
-
Contents hide
Introduction to Copper and Its Significance in Various Industries
Copper is a vital industrial metal known for its excellent ductility, electrical conductivity, and thermal conductivity. These properties make copper indispensable in social and economic development. Copper is not only abundant in nature but also possesses superior electrical conductivity, thermal conductivity, ductility, corrosion resistance, and wear resistance.
1.1 Electrical and Electronics Industry
- 1.1.1 Power Transmission: Copper is widely used in power transmission systems, such as wires and cables, transformers, switches, connectors, and terminals.
- 1.1.2 Motor Manufacturing: Copper is used in the manufacturing of stators, rotors, shaft heads, and hollow conductors in motors.
- 1.1.3 Communication Equipment: Copper wires are used in communication cables and residential electrical wiring.
- 1.1.4 Vacuum Devices: High-frequency and ultra-high-frequency transmitters, waveguides, magnetrons, and other devices require high-purity oxygen-free copper and dispersion-strengthened oxygen-free copper.
1.2 Energy and Petrochemical Industry
- 1.2.1 Thermal Power Generation: The main condenser tube sheets and condenser tubes of thermal power plants are made of brass, bronze, or cupronickel.
- 1.2.2 Solar Heaters: Copper tubes are commonly used in solar heaters.
- 1.2.3 Petrochemical Industry: Copper and various copper alloys are used to manufacture containers, pipeline systems, filters, pumps, valves, evaporators, heat exchangers, and condensers that come into contact with corrosive media.
1.3 Marine and Shipbuilding Industry
- 1.3.1 Marine Industry: Due to copper’s resistance to seawater corrosion and its bactericidal properties, copper and its alloys are widely used in desalination plants, offshore oil and gas platforms, and other coastal and underwater facilities.
- 1.3.2 Shipbuilding: Copper alloys such as aluminum bronze, manganese bronze, and aluminum brass are used to manufacture ship propellers, bolts, condenser tubes, and rivets.
1.4 Automotive and Transportation Industry
- 1.4.1 Automotive Industry: Copper and copper alloys are used in radiators, brake system pipelines, hydraulic devices, gears, bearings, distribution, and power systems.
- 1.4.2 Railways and Aviation: Electrified railways require a large amount of copper, and aircraft use copper for wiring, hydraulic, cooling, and pneumatic systems.
1.5 Construction and Mechanical Engineering
- 1.5.1 Construction Industry: Copper is used in building plumbing systems, roofing materials, and decorative items.
- 1.5.2 Mechanical Engineering: Copper is used to manufacture various transmission and fastening components such as cylinder sleeves, connectors, fasteners, and gears.
1.6 Emerging Industries
- 1.6.1 Renewable Energy: Copper plays a crucial role in solar and wind energy equipment.
- 1.6.2 High-Tech Industry: Copper is widely used in integrated circuits, semiconductors, and other high-tech devices.

-
Importance of Understanding Copper’s Melting Point
2.1 Preventing Structural Failure
When the temperature exceeds the melting point, copper transitions from solid to liquid, leading to structural failure. Therefore, it is essential to ensure that the operating temperature of copper products remains below their melting point during design and use.
2.2 Selecting Suitable Processing Methods
The melting point of copper is crucial for choosing appropriate processing methods. Knowing the melting point helps manufacturing technicians determine whether copper is suitable for specific processes such as casting, welding, and forging. For example, casting and welding processes require heating copper above its melting point for shaping and joining.
2.3 Material Selection and Design
Understanding the melting point aids engineers in selecting suitable materials and designs to ensure product safety and reliability. For instance, when using copper in electronic devices and heat exchangers, the melting point must be considered to prevent overheating and damage.
2.4 Saving Energy and Costs
Knowing the melting point of copper can help select appropriate furnaces and energy sources, thereby saving energy and reducing costs. During the processing of copper, choosing the right temperature and conditions can lower energy consumption and increase production efficiency.
2.5 Preventing Creep Failure
Creep refers to the slow deformation of materials under high temperature and stress. Copper can experience creep failure when it approaches its melting point. Understanding copper’s melting point helps operators work within safe temperature ranges to prevent creep failure.
2.6 Selecting the Appropriate Copper Alloy
| Material | Melting Point of Copper in Celsius (°C) | Melting Point of Copper in Fahrenheit (°F) | Melting Point of Copper in Kelvin (K) |
| Pure Copper (Cu) | 1,085 | 1,984 | 1,357 |
The melting point of copper alloys varies due to their different compositions. Understanding the melting points of various copper alloys can help in selecting the appropriate alloy to meet specific application needs. For example, the melting points of bronze and brass are 913°C and 930-940°C, respectively.
-
What is the Melting Point of Copper?
The melting point of copper, also known as the melting temperature, is the specific temperature at which copper transitions from a solid to a liquid state. This temperature is crucial for understanding the structural and chemical changes copper undergoes during this phase transition.
3.1 Melting Point of Pure Copper
The temperature at which pure copper begins to melt is:
3.2 Melting Point of Copper Alloys
Copper alloys are widely used in engineering, and their melting points vary depending on their composition. Here are some common copper alloys and their melting points:
| Copper Alloy | Melting Temperature of Copper Alloys (°C) |
| Arsenic copper | 685 |
| Beryllium copper | 870 to 980 |
| Brass | 930 to 940 |
| Bronze | 913 |
| Cupronickel | 1170 to 1240 |
| Chinese silver | 961 |
| Gunmetal | 900 1000 |
-
What Factors Affect the Melting Point of Copper?
Understanding the factors that influence the melting point of copper is essential for its application across various fields. These factors include alloy composition, environmental conditions, impurities, and grain size. Each aspect plays a crucial role in determining the exact melting temperature of copper. Let’s explore these factors in detail:
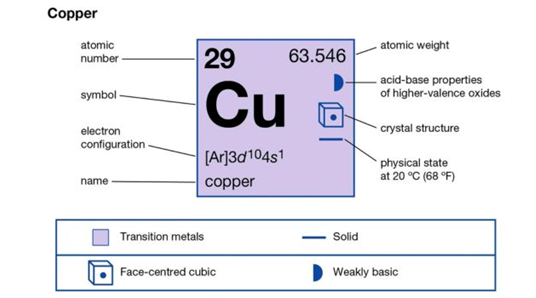
4.1 Alloy Composition
Adding other elements to form alloys significantly impacts the melting point of copper. Different alloying elements have unique melting points that can alter the overall melting point of the alloy.
- Arsenic: Lowers the melting point.
- Beryllium: Raises the melting point.
- Zinc (Brass): Lowers the melting point.
- Tin (Bronze): Lowers the melting point.
- Nickel (Cupronickel): Raises the melting point.
The specific combination and proportion of elements will determine the melting behavior of the alloy.
4.2 Environmental Factors
Environmental conditions, such as atmospheric pressure, can affect the melting point of copper.
- Pressure: Generally, increasing pressure raises the melting point of metals. At higher altitudes, where pressure is lower, the melting point of copper might be slightly reduced.
- Humidity and Ambient Temperature: These can indirectly influence the melting process by affecting the heat transfer efficiency during melting operations.
4.3 Impurities in Copper
- Impurity Effects: Impurities disrupt the orderly lattice of pure copper, making it easier for atoms to move, thus lowering the melting point.
- Phase Stability: Impurities are often more stable in the liquid phase than in the solid phase, leading to a reduction in the melting point.
4.4 Copper Grain Size
The grain size and structure of copper have profound effects on its melting point. Smaller grains typically result in a lower melting point due to several factors:
- Hall-Petch Relationship: This relationship describes how reducing grain size increases the strength and hardness of metals. However, smaller grain sizes also lower the melting point because grain boundaries influence the melting process.
- Grain Boundary Energy: Grain boundaries are areas where crystals of different orientations meet. These boundaries have higher energy compared to the interior of the grains. Smaller grains mean more grain boundaries, resulting in a lower melting point.
- Dislocation Density: This refers to the number of dislocations within the grains. Higher dislocation density typically leads to a lower melting point because dislocations can serve as sites for the initiation of melting.
- Microstructural Heterogeneity: Variations in microstructure, such as differences in grain size and composition within the material, can lead to varied melting behavior. Heterogeneous structures generally have lower melting points.
- Surface Energy: The surface energy of particles can affect the melting point. High surface energy favors the transition from solid to liquid phase, lowering the melting point.
- Diffusion Rate: Higher diffusion rates, common in fine-grained materials, lead to a lower melting point because atoms can move more freely and transition to the liquid phase more easily.
- Stress Concentration: Internal stresses within copper can lower the melting point. Stress concentration points often occur at grain boundaries or defects, acting as nucleation sites for melting and reducing the overall melting temperature.
-
Comparison of Copper’s Melting Point with Other Metals
| Metal | Melting Point of Metals and Metalloid in °C |
| Copper | 1084 |
| Melting point | 660 |
| Cast iron | 1127 to 1204 |
| Carbon steel | 1371 to 1593 |
| Chromium | 1860 |
| Cobalt | 1495 |
| Incoloy | 1390 to 1425 |
| Inconel | 1390 to 1425 |
| Stainless steel | 1510 |
| Molybdenum | 2620 |
| Magnesium | 349 to 649 |
| Niobium | 2470 |
| Nickel | 1453 |
| Tantalum | 2980 |
| Titanium | 1670 |
| Tungsten | 3400 |
| Thorium | 1750 |
| Rhenium | 3186 |
| Rhodium | 1965 |
| Ruthenium | 2482 |
| Osmium | 3025 |
| Platinum | 1770 |
| Palladium | 1555 |
| Vanadium | 1900 |
| Zirconium | 1854 |
5.1 Copper and Aluminum Melting Points
- Copper: 1,675-1,981°F / 913-1,082°C.
- Aluminum: 1,218°F / 659°C.
5.2 Copper, Gold, and Silver Melting Points
- Copper: 1,675-1,981°F / 913-1,082°C.
- Silver: 1,762°F / 961°C.
- Gold: 1,945°F / 1,063°C.
-
Methods for Melting Copper
Several methods can be used to heat copper to its melting point, such as:
- Laser Melting: High-power laser beams are used to melt copper, allowing precise control over the melting process.
- Electron Beam Melting: This technique uses high-energy electron beams to raise the temperature of copper above its melting point, suitable for achieving high purity in molten copper.
- Oxy-fuel Torch: A combination of propane and oxygen is used to produce a high-temperature flame capable of effectively melting copper.
- Furnace Heating: Various types of furnaces, including cupola furnaces, gas furnaces, and induction furnaces, can be used to melt copper. Each type has different advantages in terms of temperature control and efficiency.
- Microwave Melting: Microwave-assisted furnaces use microwave radiation to heat and melt copper, providing a modern and efficient melting method.
- Salt Bath Melting: This method uses cyanide salts and chlorides to distribute heat evenly around copper, making it ideal for processing anhydrous copper sulfate.
- Infrared Heating: Infrared heaters minimize oxidation while effectively heating copper to its melting point.
- Direct Electric Heating: This rapid and efficient method involves passing an electric current directly through copper, generating the heat required for melting.
- Vacuum Melting: A vacuum chamber is used to melt copper without contamination, allowing high temperatures while maintaining the purity of copper.
- Plasma Arc Melting: Plasma arc technology melts copper quickly and efficiently, suitable for various copper alloys.
- Casting: In processes typically used for producing complex parts, high temperatures are applied to melt copper.
- Hot Isostatic Pressing: This method combines high pressure and high temperature to melt copper materials, providing uniform performance and reducing defects.
- Cold Crucible Melting: Electromagnetic induction is used to melt copper in a water-cooled crucible, preventing contamination and allowing precise control.
- Solar Furnace: This environmentally friendly method uses solar energy to heat and melt copper, leveraging green technology for the melting process.
-
Practical Applications of Copper’s Melting Point
Understanding copper’s melting point is essential for its practical application in various industrial processes. Here, we explore how the melting point of copper is applied in casting, welding, and forging processes.
7.1 Application in Casting Processes
- Copper Castings:
-
- Relevance of Melting Point: Copper’s melting point of approximately 1,085°C (1,984°F) is a critical factor in the casting process. Knowing this temperature helps in selecting the appropriate furnaces and mold equipment to ensure efficient melting and casting of copper.
- Process Overview: During casting, copper is melted in a furnace and then poured into molds to create various shapes and objects. This process is used to produce items such as sculptures, pipe fittings, jewelry, and automotive parts.
- Equipment Used: The choice of furnaces, such as induction or crucible furnaces, is based on their ability to reach and maintain the required melting temperature for copper. Proper temperature control ensures the quality and integrity of the cast products.
7.2 Application in Welding Processes
- Copper Welding:
-
- Relevance of Melting Point: The melting point of copper is crucial in welding processes, such as brazing and soldering with copper rods. Understanding the melting point ensures that the correct temperature is reached to facilitate the bonding of copper components.
- Process Overview: In welding, copper is heated to its melting point to form a strong bond between two pieces. This process is commonly used in electrical applications, plumbing, and the manufacturing of heat exchangers and transformers.
- Techniques Used: Various welding techniques, such as arc welding, oxy-acetylene welding, and resistance welding, rely on precise temperature control to melt copper and achieve durable welds. Copper’s high thermal conductivity requires careful management of heat input to prevent overheating and ensure clean welds.
7.3 Application in Forging Processes
- Copper Forging:
-
- Relevance of Melting Point: While forging typically involves shaping metals at temperatures below their melting points, understanding copper’s melting point is still important. It helps determine the appropriate heating temperature to achieve the desired ductility without reaching the melting point.
- Process Overview: In forging, copper is heated to a temperature where it becomes malleable but does not melt. This allows it to be shaped using compressive forces generated by hammers, presses, or rolling machines. Forging enhances the mechanical properties of copper, making it stronger and more durable.
- Applications: Forged copper parts are used in various industries, including automotive, aerospace, and electrical. Common forged items include pipe fittings, electrical connectors, and heat exchangers. This process ensures that the final products have superior strength and precision.
-
Conclusion
Understanding the melting point of copper is crucial for its effective use in casting, welding, and forging processes. This knowledge ensures the application of appropriate temperatures and techniques, resulting in the production of high-quality and reliable copper products. Whether manufacturing intricate castings, robust welds, or durable forged parts, copper’s melting point plays a vital role in its practical applications across various industries.


