
Zinc plating prevents a significant portion of the $276 billion in yearly corrosion-related losses faced by US industries. The worldwide figures total nearly $1 trillion. This protective coating has become the lifeblood of manufacturing and industrial applications where metal preservation matters. Right now, zinc metal makes up one-third of worldwide production. Companies use it for galvanization, which shields metal surfaces from rust and wear.
Knowing the basic differences between zinc-plated and galvanized finishes helps make better decisions. Hot-dip galvanizing creates a thick, durable coating with a dull finish. Zinc plating needs an electrolytic solution with an electrically charged anode of pure zinc. Both methods protect well against corrosion but work differently in how they’re applied, their thickness, and how long they last. Zinc galvanizing works great because zinc costs less than other metals. This makes the plating technique more economical than other options. This piece gets into hot-dipped galvanized vs zinc-plated processes to help professionals pick the best protection method they need.
Understanding Zinc Plating and Galvanizing
Steel with zinc coating ranks among the most popular types of steel because it lasts longer and combines steel’s strength with zinc’s protection against corrosion. Zinc has protected metal from wear since 79 AD, which proves how well it works at preventing metal from breaking down.
What is zinc plating, and how does it work
Zinc plating is an electrochemical process that puts a thin zinc layer on metal surfaces. The process makes the metal substrate work as a cathode while zinc works as an anode in an electrolyte solution. When electric current flows through the bath, zinc ions move from the anode and stick to the metal substrate’s surface.
The coating creates a protective barrier – zinc breaks down about 30 times slower than untreated steel and shields the metal underneath. The whole process starts with a deep clean using alkaline detergent solutions. An acid treatment (pickling) comes next to remove surface oxides and helps the coating stick better.
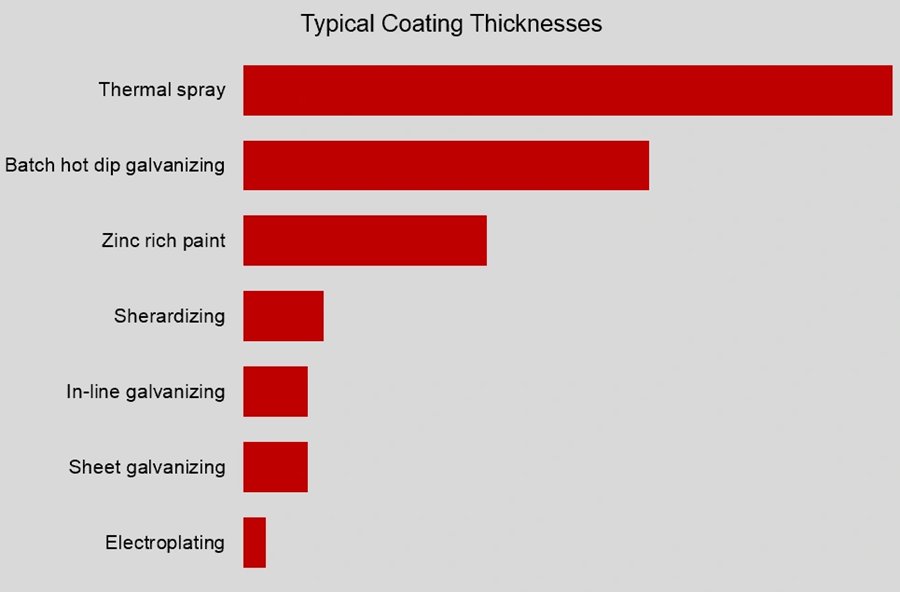
Zinc plating thickness ranges from 5μm (0.005mm) to 25μm (0.025mm). This makes it perfect for small parts like fasteners, crank handles, springs, and hardware items. Parts often go through post-treatment steps after plating. These include passivation, which can add different colors and make the coating last longer.
What is galvanizing, and how is it different
Hot-dip galvanizing dips iron or steel in molten zinc heated to about 450°C (842°F). The high temperature triggers a metallurgical reaction between the iron in steel and molten zinc. This creates a coating that covers all surfaces evenly.
The coating reacts with oxygen when it leaves the zinc bath. This creates zinc oxide, which mixes with carbon dioxide to form zinc carbonate. This protective layer gives galvanized steel its typical dull gray look. The coating has multiple layers of zinc-iron alloy and pure zinc. Its thickness usually ranges from 45μm (0.045mm) to over 100μm (0.1mm).
Hot-dip galvanizing creates a true metallurgical bond between zinc and steel, unlike zinc plating. The coating becomes part of the base metal rather than just sticking to its surface.
Zinc plated vs galvanized: key terminology explained
People often get confused about zinc coating terms. “Galvanizing” usually means hot-dip galvanizing when no other details are given. “Electro-galvanizing” means zinc plating, though these methods and their results are quite different.
These processes have several key differences:
- Appearance: Hot-dip galvanized products look dull gray and slightly rough with a crystal pattern called “spangle.” Zinc-plated items look shiny and smooth.
- Coating Thickness: Zinc plating creates a thinner layer (5-25μm) than hot-dip galvanizing (45-200μm). This affects how long they last.
- Protection Mechanism: Both methods let zinc protect steel by corroding first. Hot-dip galvanizing also heals itself – nearby zinc can still protect steel even if the coating gets damaged.
- Applications: Zinc plating works best indoors and on smaller parts. Hot-dip galvanizing shines in outdoor settings and structural projects.
These basic differences help you pick the right coating method based on your project needs and environmental conditions.
Process Comparison: Electroplating vs Hot-Dip Galvanizing
The biggest difference between these zinc coating techniques comes from how they are applied. Both methods aim to coat metal surfaces with zinc but use completely different approaches.
Electrolytic deposition in zinc plating
Zinc plating works through an electrochemical process that uses electrical current to deposit zinc particles on metal surfaces. The process starts by putting the metal part in an electrolyte solution with dissolved zinc salts. This solution can be cyanide-based, alkaline non-cyanide, or acid chloride salt formulations. The parts need thorough cleaning before they go into a barrel, rack, or drum and get submerged in the plating solution.
The metal component acts as a cathode while pure zinc serves as an anode in the solution. Electric current flowing through the system dissolves zinc from the anode and deposits it onto the cathode surface. This process happens at room temperature and creates a thin coating between 5-12 microns thick.
The type of electrolyte makes a big impact on the final result. Cyanide-based baths work well but raise environmental and safety concerns. Alkaline non-cyanide zinc solutions give better plate coverage from high to low current density areas. Adding brightness agents can boost luster, but these need careful monitoring to keep coating quality high.
Molten zinc immersion in hot-dip galvanizing
Hot-dip galvanizing takes a different approach using heat immersion. Steel components get completely submerged in molten zinc kept at 830-870°F (435-455°C). The high temperature creates a chemical reaction between the steel’s iron and the molten zinc.
The process works through diffusion, where zinc-iron alloy layers form on all surfaces, creating an even thickness throughout. The zinc bath contains at least 98% pure zinc. The coating hardens after removal from the bath and creates a strong protective layer that bonds with the base metal.
Hot-dip galvanizing creates a stronger bond than zinc plating because it chemically joins zinc and steel. This makes the coating more durable since it becomes part of the steel instead of just sticking to its surface.
Surface preparation and post-treatment differences
Both methods need proper surface preparation but follow different steps. Zinc plating requires:
- Alkaline or electrolytic cleaning to remove organic contaminants
- Pickling to remove surface oxides
- Rinsing between each step
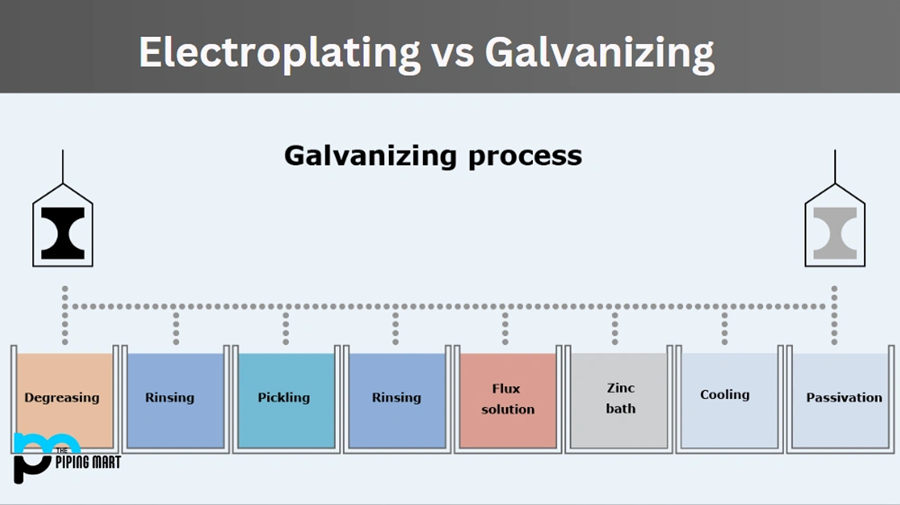
Hot-dip galvanizing needs more steps:
- Degreasing in a hot alkaline solution
- Water rinsing
- Pickling in dilute hydrochloric or sulfuric acid
- Additional water rinsing
- Fluxing in zinc ammonium chloride
Each method handles post-treatment differently. Zinc-plated parts usually get passivation treatments to last longer and might receive chromate-conversion coatings to protect against corrosion or add different translucent colors. These coatings can use either hexavalent or trivalent chromium.
Hot-dip galvanized parts might need water quenching or air cooling. The plant’s temperature, humidity, and air quality don’t affect the galvanized coating’s quality. These factors matter more for zinc plating results.
Performance Factors: Durability, Thickness, and Corrosion Resistance
The way zinc coatings perform depends on their thickness, how they’re applied, and how well they resist corrosion. These elements help determine if a coating will work well in different settings and uses.
Coating thickness: 5–25μm vs 50–200μm
A zinc coating’s durability is linked to how thick it is. Zinc plating creates thin coatings between 5μm (0.005mm) and 25μm (0.025mm). Hot-dip galvanizing, on the other hand, results in much thicker coatings – usually between 45μm (0.045mm) and over 100μm (0.1mm).
This difference in thickness plays a huge role in how long the coating lasts. The basic rule is simple: thicker coatings last longer. Hot-dip galvanizing puts about 10 times more zinc on components than zinc plating does. This is why galvanized coatings can protect metal for decades without rusting, even in tough environments.
Galvanized zinc coating in outdoor environments
Galvanized coatings work great outdoors because they’re thick and create protective compounds. The zinc surface changes when exposed to air, first forming zinc oxide. This oxide then mixes with moisture to create zinc hydroxide. These compounds then react with carbon dioxide to form a stable zinc carbonate patina.
This zinc carbonate (ZnCO3) layer acts as a protective barrier that corrodes very slowly, depending on the environment. The patina slows down zinc’s corrosion to about 1/30th the rate of steel in similar conditions. For outdoor uses like structural steel, farm equipment, and fencing, hot-dip galvanizing stands out as the best choice.
Zinc plating for indoor and cosmetic applications
Zinc plating works well enough for parts used in mild or indoor settings. The thinner coating suits small fasteners, light switch plates, hardware parts, and items that won’t face harsh conditions. These coatings aren’t meant for outdoor use because they’re too thin.
Zinc plating looks great, which makes it perfect for applications where appearance counts. The coating can look different – from dull gray with a matte finish to brighter, more shiny finishes based on the process or additives used. It’s a cost-effective solution for parts that need to look good while offering decent corrosion protection.
Sacrificial protection and zinc carbonate formation
Both methods protect steel through cathodic protection – zinc corrodes first to protect the steel underneath. If the coating gets damaged and exposes bare steel, the zinc sacrifices itself to protect the steel. This happens because zinc is more electron-donating than carbon steel.
Hot-dip galvanized coatings have an interesting feature – even if damage exposes steel up to about ¼ inch wide, rust won’t start until the surrounding zinc is gone. The zinc carbonate that forms on the surface adds ongoing protection. The patina takes over a year to fully develop and extends the coating’s life significantly. This self-healing ability makes galvanized coatings special – scratches often fill in with zinc oxides and carbonates, which helps stop further corrosion.
Cost, Safety, and Environmental Considerations
The choice of zinc coating method depends on both economic and environmental factors, beyond just performance characteristics.
Material and equipment costs for each method
Zinc plating equipment works at room temperature and needs less energy but more chemicals than hot-dip galvanizing. Hot-dip galvanizing equipment costs more but creates thicker coatings faster. The original setup costs for galvanizing are higher, though it becomes more cost-effective over time. Zinc price variations between $1.04-$1.64/lb substantially affect both processes. Galvanizers charge $0.20-$0.50 per pound of finished weight, but lose $0.69-$0.99 for each extra pound of zinc beyond what’s required.
Toxicity and disposal: cadmium vs zinc vs galvanizing
Cadmium electroplating provided excellent corrosion protection until its toxic nature became clear. While zinc plating eliminated cadmium’s cancer risks, some zinc plating processes use chromate with potential health risks. The UK’s hot-dip galvanizing industry produces about 32,000 tons of acid waste yearly, with disposal costs around £40/ton. This waste contains valuable materials – a 40m³ acid tank with 130g/l iron concentration holds 5.2 tons of iron that usually ends up in landfills.
Eco-friendliness and recyclability of zinc galvanizing
Zinc and steel can be recycled endlessly without losing their physical or chemical properties. Mined ores supply 70% of zinc while recycled sources provide the remaining 30%. The recovery and recycling rate for available zinc exceeds 80%. A strong recycling system helps reduce energy use, emissions, and waste. Galvanized products last 50-100 years, and builders recover 95% of zinc products from structures at the end of their lifecycle.
Choosing the Right Method for Your Application
Choosing between zinc plating and hot-dip galvanizing depends on your application’s specific needs. These methods shine in different scenarios, and each has its own advantages.
Hot-dipped galvanized vs zinc-plated for automotive parts
Car manufacturers heavily depend on galvanized steel. The “body-in-white” makes up about 80% of car bodies. Galvanized coating’s durability in outdoor environments makes it a top choice. Zinc plating works best for critical components like brake calipers, pipes, and power steering parts. It protects these parts while keeping their precise measurements intact.
When to use zinc plating for small hardware
Small, detailed components look great with zinc plating. This method works perfectly for nuts, bolts, washers, and indoor hardware items. It combines cost-effectiveness with visual appeal. The bright, metallic finish makes these items look great in consumer products. Zinc plating’s thin coating helps fasteners maintain precise threading, so you won’t need to overtap nuts.
Galvanizing for structural steel and outdoor use
Most structural steel projects follow ASTM A123/123M standards for hot-dip galvanizing. Galvanized coatings can fix minor damage by themselves. They react with air to create protective zinc carbonate. This self-healing feature makes them perfect for bridges, farm equipment, and outdoor structures that face harsh weather.
Avoiding zinc plating in high-temperature environments
Heat poses a major challenge for both coatings. Zinc plating starts to break down at temperatures above 212°F. You should avoid using zinc plating in any application that reaches over 500°F. Hot-dip galvanizing also has its limits – it’s not suitable for highly acidic environments or places where temperatures exceed 400°F.
Conclusion
Zinc-based coatings provide vital protection against corrosion and save billions of dollars yearly in industries of all sizes. The comparison between zinc plating and hot-dip galvanizing reveals notable differences in how they’re applied, their coating thickness, and durability.
Zinc plating uses an electrolytic process that creates thinner coatings (5-25μm) and works well for components used in mild or indoor environments. Small hardware items, fasteners with precise threading, and projects where looks matter benefit from this method. Hot-dip galvanizing creates much thicker coatings (45-200μm) through metallurgical bonding. This makes it perfect for structural steel, outdoor structures, and equipment that faces harsh conditions.
These methods shield the underlying steel as zinc corrodes first to protect it. Hot-dip galvanizing goes further by offering self-healing properties through zinc carbonate formation. This extends its service life in tough environments. The patina that develops explains why galvanized coatings last for decades without rust appearing.
The costs vary based on what you need. Zinc plating equipment runs at room temperature and needs less energy but more chemicals. Hot-dip galvanizing costs more to set up but saves money over time because it lasts longer and needs less maintenance.
The environmental impact plays a role in choosing between methods. Both zinc and steel can be recycled endlessly without losing their properties. People recover and reuse about 80% of available zinc, making these processes more eco-friendly than other options.
Your specific needs determine whether zinc plating or hot-dip galvanizing works better. Projects that need exact measurements, good looks, and indoor use do well with zinc plating. You’ll want hot-dip galvanizing for structural parts, outdoor use, and items that face harsh conditions. Both methods have their limits – neither works well in extreme heat above 400-500°F.
Engineers and manufacturers who know these basic differences can pick the right coating method based on their project’s needs, environment, and how long it needs to last. Zinc-based coatings protect valuable metal assets from wear and tear, no matter which method you choose. They extend service life and keep everything working as it should.
Key Takeaways
Understanding the fundamental differences between zinc plating and galvanizing helps manufacturers select the optimal corrosion protection method for their specific applications and environmental conditions.
• Coating thickness determines durability: Zinc plating creates thin coatings (5-25μm) suitable for indoor use, while galvanizing produces thick coatings (45-200μm) ideal for outdoor applications.
• Process differences impact performance: Zinc plating uses electrochemical deposition at room temperature, while galvanizing involves molten zinc immersion at 450°C, creating metallurgical bonds.
• Application-specific selection is crucial: Choose zinc plating for small hardware, fasteners, and esthetic applications; select galvanizing for structural steel and harsh outdoor environments.
• Both methods provide sacrificial protection: Zinc corrodes preferentially to protect underlying steel, but galvanizing offers additional self-healing through zinc carbonate formation.
• Cost considerations vary by application: Zinc plating has lower setup costs but galvanizing proves more economical long-term due to superior durability and reduced maintenance needs.
The key to successful corrosion protection lies in matching the coating method to your specific requirements – considering factors like environment, durability needs, appearance, and long-term cost effectiveness.
FAQs
Q1. Which coating method provides better protection: zinc plating or galvanizing? Hot-dip galvanizing generally provides superior protection, especially for outdoor applications. It creates a thicker coating (45-200μm) that is metallurgically bonded to the steel, offering better durability and corrosion resistance compared to the thinner zinc plating (5-25μm).
Q2. How long do zinc plated and galvanized coatings typically last? Galvanized coatings typically last much longer than zinc plating. Hot-dip galvanized items can remain functional for 50-100 years in many environments, while zinc plated items are better suited for indoor use and have a shorter lifespan, especially in harsh conditions.
Q3. What are the main drawbacks of zinc plating? Zinc plating has limited corrosion resistance in wet or high-humidity environments. It’s also chemically susceptible to acids and alkalis. The thin coating may not provide sufficient protection for outdoor use, and there’s a potential risk of hydrogen embrittlement during the plating process.
Q4. Which coating is more suitable for outdoor applications? Hot-dip galvanizing is significantly more suitable for outdoor applications. Its thicker zinc coating provides better rust protection in wet climates and harsh outdoor environments. Zinc plating is more appropriate for indoor use or in dry climates where only superficial protection is needed.
Q5. How do zinc plating and galvanizing differ in terms of cost and environmental impact? Zinc plating generally has lower initial costs but may require more frequent reapplication. Galvanizing has higher upfront costs but often proves more economical long-term due to its durability. Both methods use recyclable materials, with over 80% of available zinc being recovered and recycled, making them relatively eco-friendly options.
